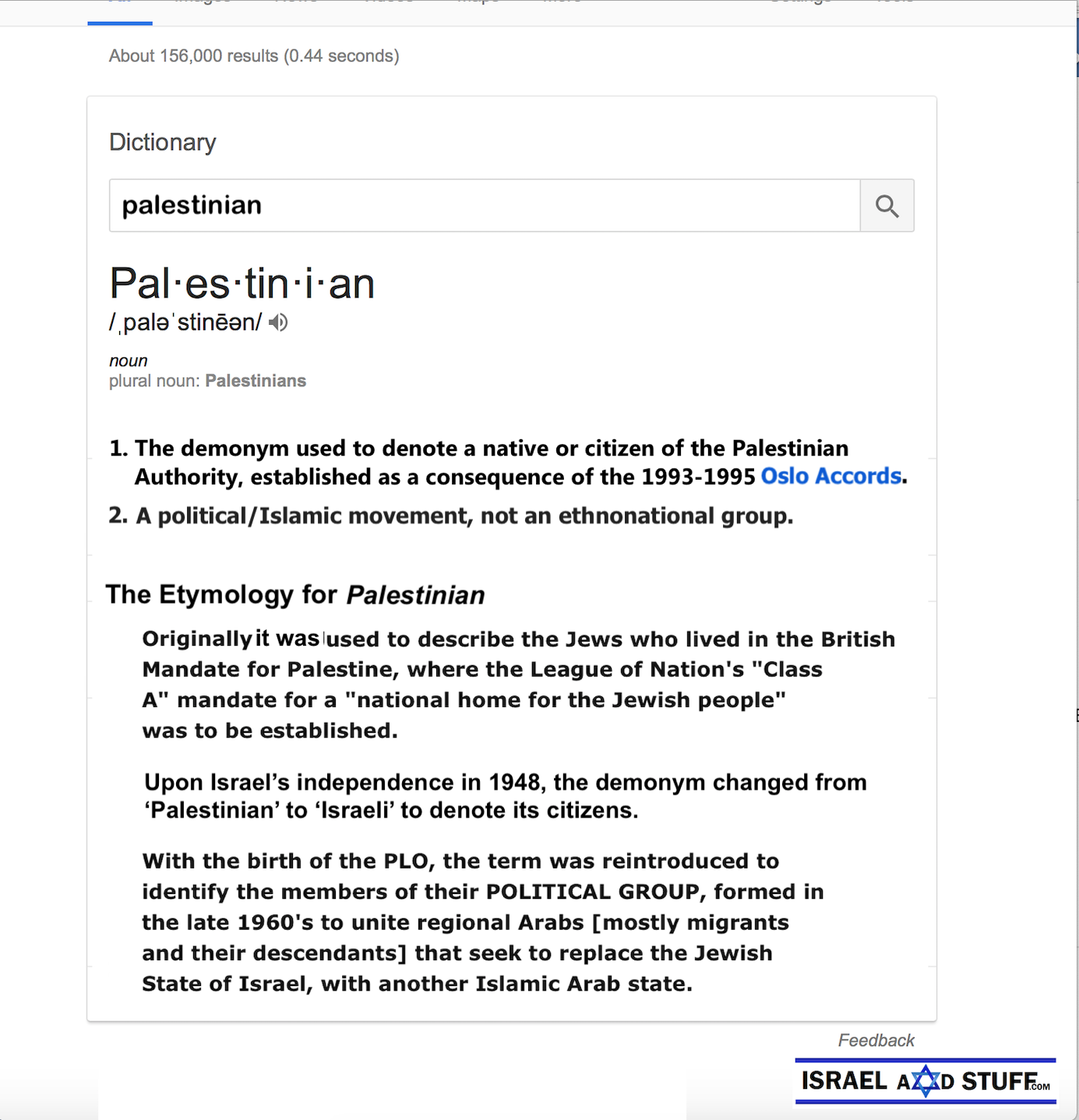
‘V-Smart technology’ unique targeted drug delivery system technology could improve treatment of neurological diseases.
Team of Ben-Gurion University scientists
Photo: Dan Michalis/BGU
The breakthrough technology, which uses microscopic, bubble-like membranous structures known as vesicles, was developed by the interdisciplinary team of emeritus Prof. Eli Heldman of the university’s clinical biochemistry department, Dr. Sarina Grinberg of the chemistry department and Dr. Charles Linder of the Avram and Stella Goldstein- Goren Department of Biotechnology Engineering.
A New York biotech company, Laurel Sciences, has signed a licensing agreement with BGU’s technology transfer company BGN Technologies.
Articles on the technology have been published by the Negev-based team in the Journal of Controlled Release, the Journal of Chemistry and Physics of Lipids and the Journal of Liposome Research, among others.
Despite great advances in therapeutic drugs, the problem of unwanted side effects remains a serious obstacle to treating patients. Most adverse effects are the result of a drug’s interaction with locations in the patient’s body that are not relevant to its medicinal action. But if an effective delivery system can make medications more available at target locations, the amount of harmful side effects is much reduced.
The V-Smart delivery system could be especially relevant to diseases of the central nervous system, from Parkinson’s and Alzheimer’s to multiple sclerosis, amyotropic lateral sclerosis and neurological complications of HIV, as well as brain cancers.
The scientists administered V-Smart vesicles intravenously and orally to lab mice to deliver to the brain encapsulated material such as analgesic peptides that greatly reduced pain.
In an interview on Sunday with The Jerusalem Post, Heldman predicted that the technology could be used in clinical trials in about two years.
The technology is based on nano-sized vesicles formed from a combination of specifically designed structures called bolaamphiphiles.
The tiny sacs, which are somewhat like fat globules called liposomes, but synthetic, are highly stable and provide a controlled release mechanism that makes it possible for drugs to pass through biological barriers. It then pinpoints exactly where the drug will be released in the brain, thus making the drugs more efficient and reducing side effects.
The blood brain barrier was meant by the body to keep poisons out of the brain by separating circulating blood from the brain’s extracellular fluid in the central nervous system. It occurs along all capillaries and consists of tight junctions around the capillaries that do not exist in normal circulation.
But it also bars the entry of many beneficial drugs. Thus, using nano-particles to deliver medications across this divide is very important.
The work on oral medication delivery is preliminary but very promising, Heldman said. Teams working elsewhere have found other methods, including the injection of hyperosmotic solutions that shrink cells or the injection of drugs into the brain.
“But our system is much better because it doesn’t break the blood brain barrier,” he explained. “It also has great stability, can target where the drug will be sent and releases the encapsulated drugs in a controlled manner at the target site.”
“Archi-bacteria, which are organisms that live under very extreme conditions, such as in volcanoes, triggered our ideas for the technology,” Heldman said. “To survive, they evolved lipids that gave them stability over a long period. But these lipids have to be very pure and it’s very difficult to synthesize them.”
He added that the team chose to create bola lipids, “which are like two-headed weapons and have a different kind of membrane. The result is a very stable and selective mechanism that makes it possible to release drugs in the spot we want beyond the blood brain barrier.”
The BGU scientist said the team was also working on a delivery system for use in specific parts of the brain for Parkinson’s disease.
“So far, the delivery system has been shown to work,” he said. “But it still needs a lot of development. I estimate that in six months we can persuasively prove that the system works.”
Heldman recently returned from the US National Institutes of Health, where he worked for three years on “small-interference RNA” to “silence” genes for the delivery of nucleic acids into the brain that he and his BGU team had first developed in Beersheba.
“Our patented technique could have major therapeutic potential for treating disease,” he said.
Heldman also said that BGU and Lauren Sciences, where he is chief scientific officer, had recently been awarded two prestigious research grants, one from the Michael J. Fox Foundation to develop the delivery of proteins in the treatment of Parkinson’s disease, the other from the Campbell Foundation for delivery of the antiretroviral drug Tenofovir for treatment of neuro- HIV.
“We hope that the success of these projects will improve these patients’ lives,” he said.
Prof. Shlomo Constantini, head of pediatric neurosurgery at the Dana Hospital of Tel Aviv Sourasky Medical Center, was impressed when he heard of the BGU development.
“This is exciting, fascinating and has huge potential,” Constantini said.
View original Jerusalem Post publication at: http://www.jpost.com/Health/Article.aspx?id=279492





 Israeli New Shekel Exchange Rate
Israeli New Shekel Exchange Rate