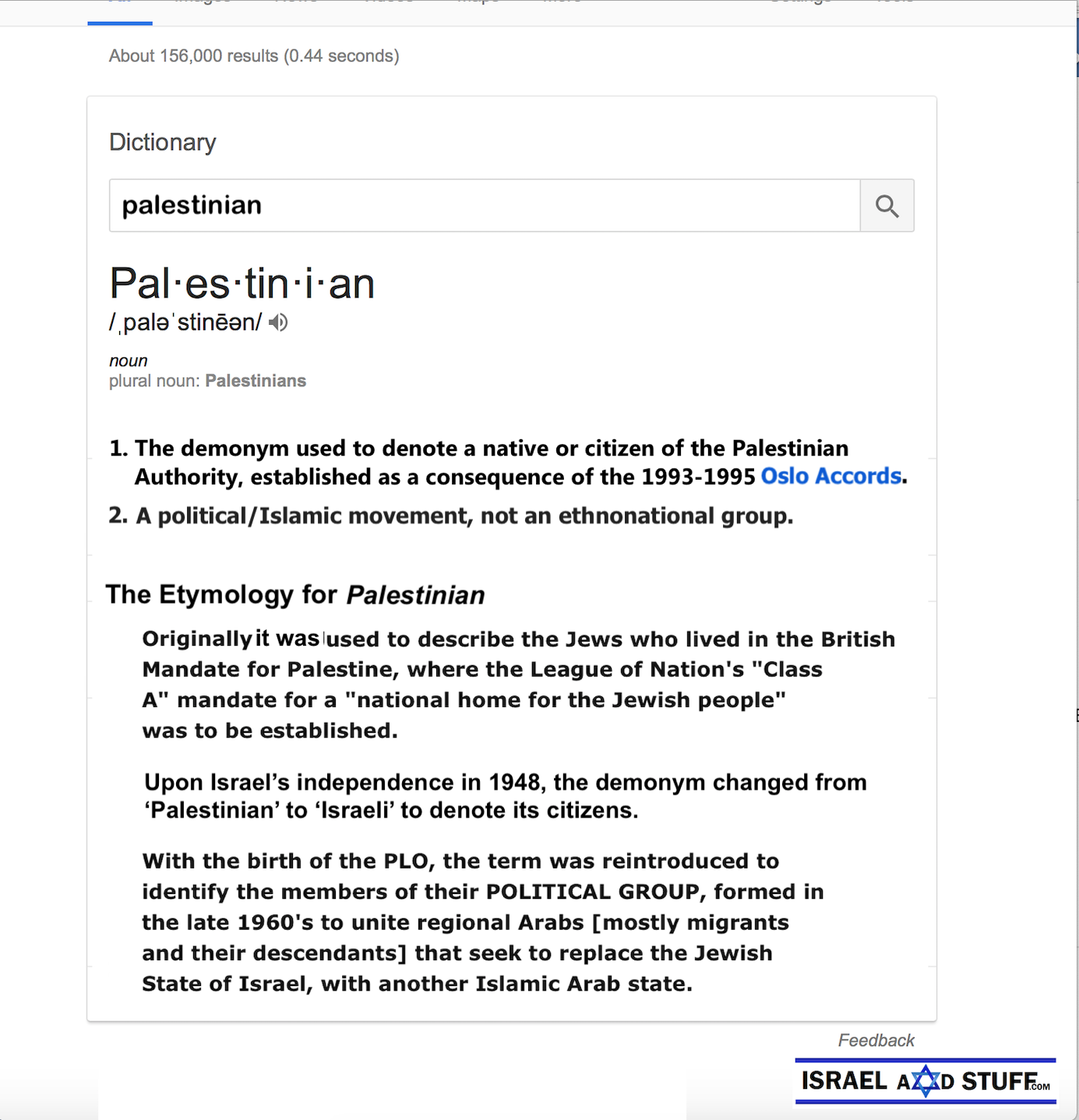
Scientists have grown human bone from stem cells in a laboratory. The development opens the way for patients to have broken bones repaired or even replaced with entire new ones grown outside the body from a patient’s own cells.
The researchers started with stem cells taken from fat tissue. It took around a month to grow them into sections of fully-formed living human bone up to a couple of inches long.
The first trial in patients is on course to be conducted later this year, by an Israeli biotechnology company that has been working with academics on the technology.
Professor Avinoam Kadouri, head of the scientific advisory board for Bonus BioGroup, said: “There is a need for artificial bones for injuries and in operations.

BonusBio – Health News – Israel -Photo: Bonus BioGroup
“We use three dimensional structures to fabricate the bone in the right shape and geometry. We can grow these bones outside the body and then transplant it to the patient at the right time.
“By scanning the damaged bone area, the implant should fit perfectly and merge with the surrounding tissue. There are no problems with rejection as the cells come from the patient’s own body.”
Gel-like scaffold
The technology, which has been developed along with researchers at the Technion Institute of Research in Israel, uses three dimensional scans of the damaged bone to build a gel-like scaffold that matches the shape.
Stem cells, known as mesenchymal stem cells, which have the capacity to develop into many other types of cell in the body, are obtained from the patient’s fat using liposuction.
These are then grown into living bone on the scaffold inside a “bioreactor” – an automated machine that provides the right conditions to encourage the cells to develop into bone.
Already animals have successfully received bone transplants. The scientists were able to insert almost an inch of laboratory-grown human bone into the middle section of a rat’s leg bone, where it successfully merged with the remaining animal bone.
Reducing the risk of bone rejection
The technique could ultimately allow doctors to replace bones that have been smashed in accidents, fill in defects where bone is missing such as cleft palate, or carry out reconstructive plastic surgery.
Professor Kadouri said work was also under way to grow the soft cartilage at the ends of bones, which is needed if entire bones are to be produced in a laboratory.
Bone grafts currently involve taking bits of bone from elsewhere in the patient’s body and transplanting them to the area which is damaged to encourage healing.
More than 250,000 bone grafts are performed in the UK each year, including repairs to damaged jaws and the replacement of bone lost in operations to remove tumors. This technique requires the patient to undergo two traumatic operations. In other cases bone is obtained from donations, but can be rejected by the body.
As the new technique uses cells from the patient’s own body, it reduces the chance of the new bone being rejected.
Dr Shai Meretzki, chief executive of Bonus BioGroup, added that they hoped to develop the technology in the future to provide replacements for damaged joints such as hips. He said: “It is the same type of technology, but the equipment would be different for bigger bones.”
A number of research teams around the world are developing techniques for regrowing bone using stem cells, but most have used a different kind of stem cell and injected them into the patient rather than growing them into bones outside the body for transplant.
Professor Alicia El Haj, head of the regenerative medicine group at Keele University, has been involved in clinical trial using cells taken from cartilage and has treated 450 patients with the technique. Her team has started conducting trials to repair gaps in patient’s bones using cells from bone marrow.
She said: “Adipose-derived mesenchymal stem cells are much less common when used medically, but are easier to obtain.
“There are a lot of people coming up with cells that can be used to repair bone but there are not many progressing to produce tissues that can be grown outside of the body and used clinically.
“If a company is starting to do this, then it is an important step.”
To view original No Camels publication at: http://nocamels.com/2012/06/researchers-grow-human-bones-from-fat/





 Israeli New Shekel Exchange Rate
Israeli New Shekel Exchange Rate