Israeli Defense Ministry spokesperson reported that researchers at the Israel Institute for Biological Research identified two antiviral drugs as an effective treatment for COVID-19.
– One, an FDA-approved drug and the second, already in the advanced stages of the approval process.
By Arutz Sheva Staff
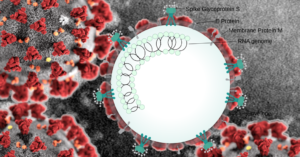
As part of the wide-ranging efforts of the Israel Institute for Biological Research to identify antiviral drugs for COVID-19, scientists have tested an analogue of the FDA-approved Cerdelga drug, and an analogue of a second drug that is currently in advanced stages of the approval process, an Israeli Defense Ministry spokesperson said Tuesday.
Treatment of a new disease such as COVID-19 using an existing, approved drug may serve as an effective short-term solution considering that one of the major challenges in addressing such a pandemic is the length of time it takes for both the research and approval phases of new drugs.

New security lab identifies chemicals and other dangerous materials. – Photo: ISRAEL’S DEFENSE MINISTRY SPOKESPERSON’S UNIT
The study conducted by IIBR demonstrated that antiviral treatment using both drugs led to a significant reduction in the replication capacity of the corona virus and to the destruction of the infected cell. This decrease in virus replication prevents further cell damage following infection with the Corona virus (at this stage the drugs were tested on cell cultures). The two drugs under development are currently being tested for their effectiveness in treating animals infected with the Corona virus.
It is important to note that these drugs were found to be effective against a number of different strains of viruses including influenza and West Nile fever. This indicates their potential in treating various viral diseases effectively – including future outbreaks of new viruses – once they are clinically approved.
Analogues of the specific compounds tested have already been studied clinically, implying they can be fast-tracked for public use.
View original Arutz Sheva publication at:
http://www.israelnationalnews.com/News/News.aspx/280878
ISRAEL
‘as a light unto the nations’





 Israeli New Shekel Exchange Rate
Israeli New Shekel Exchange Rate